Abstract
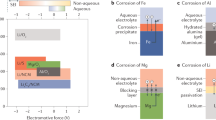
Rechargeable lithium (Li) metal batteries must have long cycle life and calendar life (retention of capacity during storage at open circuit). Particular emphasis has been placed on prolonging the cycle life of Li metal anodes, but calendar ageing is less understood. Here, we show that Li metal loses at least 2–3% of its capacity after only 24 hours of ageing, regardless of the electrolyte chemistry. These losses of capacity during calendar ageing also shorten the cycle life of Li metal batteries. Cryogenic transmission electron microscopy shows that chemical corrosion of Li and the continuous growth of the solid electrolyte interphase—a passivation film on Li—cause the loss of capacity. Electrolytes with long cycle life do not necessarily form a solid electrolyte interphase with more resistance to chemical corrosion, so functional electrolytes must simultaneously minimize the rate of solid electrolyte interphase growth and the surface area of electrodeposited Li metal.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets analysed and generated during the current study are included in the paper and its Supplementary Information file.
References
Xu, W. et al. Lithium metal anodes for rechargeable batteries. Energy Environ. Sci. 7, 513–537 (2014).
Lin, D., Liu, Y. & Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 12, 194–206 (2017).
Fang, C., Wang, X. & Meng, Y. S. Key issues hindering a practical lithium-metal anode. Trends Chem. 1, 152–158 (2019).
Fang, C. et al. Quantifying inactive lithium in lithium metal batteries. Nature 572, 511–515 (2019).
Zachman, M. J., Tu, Z., Choudhury, S., Archer, L. A. & Kourkoutis, L. F. Cryo-STEM mapping of solid–liquid interfaces and dendrites in lithium-metal batteries. Nature 560, 345–349 (2018).
Li, Y. et al. Correlating structure and function of battery interphases at atomic resolution using cryoelectron microscopy. Joule 2, 2167–2177 (2018).
Li, Y. et al. Atomic structure of sensitive battery materials and interfaces revealed by cryo–electron microscopy. Science 358, 506–510 (2017).
Wang, X. et al. New insights on the structure of electrochemically deposited lithium metal and its solid electrolyte interphases via cryogenic TEM. Nano Lett. 17, 7606–7612 (2017).
Lin, D. et al. Layered reduced graphene oxide with nanoscale interlayer gaps as a stable host for lithium metal anodes. Nat. Nanotechnol. 11, 626–632 (2016).
Cao, X. et al. Monolithic solid–electrolyte interphases formed in fluorinated orthoformate-based electrolytes minimize Li depletion and pulverization. Nat. Energy 4, 796–805 (2019).
Weber, R. et al. Long cycle life and dendrite-free lithium morphology in anode-free lithium pouch cells enabled by a dual-salt liquid electrolyte. Nat. Energy 4, 683–689 (2019).
Niu, C. et al. Self-smoothing anode for achieving high-energy lithium metal batteries under realistic conditions. Nat. Nanotechnol. 14, 594–601 (2019).
Gao, Y. et al. Polymer–inorganic solid–electrolyte interphase for stable lithium metal batteries under lean electrolyte conditions. Nat. Mater. 18, 384–389 (2019).
Adams, B. D., Zheng, J., Ren, X., Xu, W. & Zhang, J. G. Accurate determination of Coulombic efficiency for lithium metal anodes and lithium metal batteries. Adv. Energy Mater. 8, 1702097 (2017).
Crozier, C., Apostolopoulou, D. & Mcculloch, M. Clustering of usage profiles for electric vehicle behaviour analysis. In 2018 IEEE PES Innovative Smart Grid Technologies Conference Europe (ISGT-Europe) 1–6 (IEEE, 2018).
Speidel, S. & Bräunl, T. Driving and charging patterns of electric vehicles for energy usage. Renew. Sustain. Energy Rev. 40, 97–110 (2014).
Kolesnikov, A. et al. Galvanic corrosion of lithium-powder-based electrodes. Adv. Energy Mater. 10, 2000017 (2020).
Lin, D. et al. Fast galvanic lithium corrosion involving a Kirkendall-type mechanism. Nat. Chem. 11, 382–389 (2019).
Keil, P. et al. Calendar aging of lithium-ion batteries I. Impact of the graphite anode on capacity fade. J. Electrochem. Soc. 163, A1872–A1880 (2016).
Dubarry, M., Qin, N. & Brooker, P. Calendar aging of commercial Li-ion cells of different chemistries – a review. Curr. Opin. Electrochem. 9, 106–113 (2018).
Attia, P. M., Das, S., Harris, S. J., Bazant, M. Z. & Chueh, W. C. Electrochemical kinetics of SEI growth on carbon black: part I. Experiments. J. Electrochem. Soc. 166, 97–106 (2019).
Moretti, A. et al. A comparison of formation methods for graphite//LiFePO4 cells. Batter. Supercaps 2, 240–247 (2019).
Qian, J. et al. High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015).
Yu, Z. et al. Molecular design of electrolyte solvents enabling energy-dense and long-cycling lithium metal batteries. Nat. Energy 5, 526–533 (2020).
Liu, Y. et al. Solubility-mediated sustained release enabling nitrate additive in carbonate electrolytes for stable lithium metal anode. Nat. Commun. 9, 3656 (2018).
Peled, E. & Menkin, S. Review—SEI: past, present and future. J. Electrochem. Soc. 164, A1703–A1719 (2017).
Genovese, M. et al. Hot formation for improved low temperature cycling of anode-free lithium metal batteries. J. Electrochem. Soc. 166, A3342–A3347 (2019).
Wang, J. et al. Improving cyclability of Li metal batteries at elevated temperatures and its origin revealed by cryo-electron microscopy. Nat. Energy 4, 664–670 (2019).
Wang, H. et al. Wrinkled graphene cages as hosts for high-capacity Li metal anodes shown by cryogenic electron microscopy. Nano Lett. 19, 1326–1335 (2019).
Pei, A., Zheng, G., Shi, F., Li, Y. & Cui, Y. Nanoscale nucleation and growth of electrodeposited lithium metal. Nano Lett. 17, 1132–1139 (2017).
Huang, W. et al. Evolution of the solid–electrolyte interphase on carbonaceous anodes visualized by atomic-resolution cryogenic electron microscopy. Nano Lett. 19, 5140–5148 (2019).
Huang, W. et al. Dynamic structure and chemistry of the silicon solid-electrolyte interphase visualized by cryogenic electron microscopy. Matter 1, 1232–1245 (2019).
Pinson, M. B. & Bazant, M. Z. Theory of SEI formation in rechargeable batteries: capacity fade, accelerated aging and lifetime prediction. J. Electrochem. Soc. 160, A243–A250 (2013).
Zhou, Y. et al. Real-time mass spectrometric characterization of the solid–electrolyte interphase of a lithium-ion battery. Nat. Nanotechnol. 15, 224–230 (2020).
Lu, P., Li, C., Schneider, E. W. & Harris, S. J. Chemistry, impedance, and morphology evolution in solid electrolyte interphase films during formation in lithium ion batteries. J. Phys. Chem. C 118, 896–903 (2014).
Huang, W., Wang, H., Boyle, D. T., Li, Y. & Cui, Y. Resolving nanoscopic and mesoscopic heterogeneity of fluorinated species in battery solid-electrolyte interphases by cryogenic electron microscopy. ACS Energy Lett. 5, 1128–1135 (2020).
Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 104, 4303–4417 (2004).
Brown, Z. L., Jurng, S., Nguyen, C. C. & Lucht, B. L. Effect of fluoroethylene carbonate electrolytes on the nanostructure of the solid electrolyte interphase and performance of lithium metal anodes. ACS Appl. Energy Mater. 1, 26–31 (2018).
Cody, G. D. et al. Quantitative organic and light‐element analysis of comet 81P/Wild 2 particles using C‐, N‐, and O‐μ‐XANES. Meteorit. Planet. Sci. 43, 353–365 (2008).
le Guillou, C., Bernard, S., de la Pena, F. & le Brech, Y. XANES-based quantification of carbon functional group concentrations. Anal. Chem. 90, 8379–8386 (2018).
Braun, A., Kubatova, A., Wirick, S. & Mun, S. B. Radiation damage from EELS and NEXAFS in diesel soot and diesel soot extracts. J. Electron Spectros. Relat. Phenom. 170, 42–48 (2009).
Endo, E., Ata, M., Sekai, K. & Tanaka, K. Spin trapping study of gradual decomposition of electrolyte solutions for lithium secondary batteries. J. Electrochem. Soc. 146, 49–53 (1999).
Gourdin, G., Collins, J., Zheng, D., Foster, M. & Qu, D. Spectroscopic compositional analysis of electrolyte during initial SEI layer formation. J. Phys. Chem. C 118, 17383–17394 (2014).
Soto, F. A. et al. Formation and growth mechanisms of solid-electrolyte interphase layers in rechargeable batteries. Chem. Mater. 27, 7990–8000 (2015).
Huang, W. et al. Nanostructural and electrochemical evolution of the solid-electrolyte interphase on CuO nanowires revealed by cryogenic electron microscopy and impedance spectroscopy. ACS Nano 13, 737–744 (2019).
Boyle, D. T. et al. Transient voltammetry with ultramicroelectrodes reveals the electron transfer kinetics of lithium metal anodes. ACS Energy Lett. 5, 701–709 (2020).
Wood, S. M. et al. Predicting calendar aging in lithium metal secondary batteries: the impacts of solid electrolyte interphase composition and stability. Adv. Energy Mater. 8, 1801427 (2018).
Attia, P. M., Cheuh, W. C. & Harris, S. J. Revisiting the t0.5 dependence of SEI growth. J. Electrochem. Soc. 167, 090535 (2020).
Aurbach, D. & Zaban, J. Impedance spectroscopy of lithium electrodes: part 1. General behavior in propylene carbonate solutions and the correlation to surface chemistry and cycling efficiency. J. Electroanal. Chem. 348, 155–179 (1993).
Aurbach, D. & Zaban, J. Impedance spectroscopy of lithium electrodes: part 2. The behaviour in propylene carbonate solutions — the significance of the data obtained. J. Electroanal. Chem. 367, 15–25 (1994).
Aurbach, D. & Zaban, J. Impedance spectroscopy of lithium electrodes: part 3. The importance of Li electrode preparation. J. Electroanal. Chem. 365, 41–45 (1994).
Zhuo, Z. et al. Breathing and oscillating growth of solid-electrolyte-interphase upon electrochemical cycling. Chem. Commun. 54, 814–817 (2018).
Zheng, J. et al. Physical orphaning versus chemical Instability: is dendritic electrodeposition of Li fatal? ACS Energy Lett. 4, 1349–1355 (2019).
Harlow, J. E. et al. A wide range of testing on an excellent lithium-ion cell chemistry to be used as benchmarks for new battery technologies. J. Electrochem. Soc. 166, A3031–A3044 (2019).
Zaban, A., Zinigrad, E. & Aurbach, D. Impedance spectroscopy of lithium electrodes. 4. A general simple model of the Li–solution interphase in polar aprotic systems. J. Phys. Chem. 100, 3089–3101 (1996).
Acknowledgements
We acknowledge support from the Assistant Secretary for Energy Efficiency and Renewable Energy, Office of Vehicle Technologies of the US Department of Energy under the Battery Materials Research (BMR) Program and Battery 500 Consortium. The cryo-TEM research is supported by the Department of Energy, Office of Basic Energy Sciences, Division of Materials Science and Engineering under contract DE-AC02-76SF00515. D.T.B. acknowledges support from the National Science Foundation Graduate Research Fellowship Program. Scanning electron microscopy and TEM were performed at the Stanford Nano Shared Facilities (SNSF), supported by the National Science Foundation under award ECCS-1542152. The K3 IS camera and support are courtesy of Gatan. We also acknowledge H. Wang for help designing the schematic in Fig. 5.
Author information
Authors and Affiliations
Contributions
D.T.B. and Y.C. conceived the idea. D.T.B. designed the research with guidance from Y.C., carried out the electrochemical measurements and analysed the data. W.H. and Y.L. helped design the cryo-(S)TEM experiments, and W.H. carried out the cryo-(S)TEM experiments and analysed the data. W.Z. carried out the cryo-TEM selected area electron diffraction (SAED) and additional EELS measurements. H.W. and H.C. helped carry out and interpret the XPS experiments and synthesized host materials. H.C. helped with scanning electron microscopy. Z.Y. synthesized electrolyte materials. D.T.B., W.H. and Y.C. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Energy thanks Marian Stan, Yifei Yuan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–21, Tables 1–2 and references.
Rights and permissions
About this article
Cite this article
Boyle, D.T., Huang, W., Wang, H. et al. Corrosion of lithium metal anodes during calendar ageing and its microscopic origins. Nat Energy 6, 487–494 (2021). https://doi.org/10.1038/s41560-021-00787-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-021-00787-9
This article is cited by
-
Resting restores performance of discharged lithium-metal batteries
Nature (2024)
-
Recovery of isolated lithium through discharged state calendar ageing
Nature (2024)
-
Spontaneous electrochemical uranium extraction from wastewater with net electrical energy production
Nature Water (2023)
-
Nonintrusive thermal-wave sensor for operando quantification of degradation in commercial batteries
Nature Communications (2023)
-
A corrosion inhibiting layer to tackle the irreversible lithium loss in lithium metal batteries
Nature Communications (2023)